PREV ARTICLE
NEXT ARTICLE
FULL ISSUE
PREV FULL ISSUE
BOB VLACK, A PERSONAL REMEMBRANCE
Jeff Rock submitted these recollections of the late Robert Vlack. Thanks! -Editor
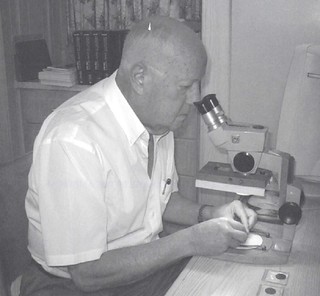
It's hard for me to believe that I first got in contact with Bob four decades ago! What is even harder to fathom is that I am now a half-century old (edging in on antique status myself), and that I was all of 10 years old when I got in touch with one of the leading researchers, writers and dealers. Harder still to imagine is Bob's response to a precocious kid – nothing but kindness and support. Bob was one of the few dealers who never "talked down" to a kid, and that made all the difference to a budding numismatist. Instead of rolling his eyes and saying "I have some nice VF Lincoln pennies to show you" he embraced the fact that a kid could have an interest in colonials – and be serious about them even when his budget was limited to birthday gift cash, allowance and paper route money. I finally met him in person in my late teens (I think it was at a Cherry Hill, New Jersey show in the early 1980's, though I am sure he was in attendance at the Garrett sales in the 1979-1980 period, though I was there just as an observer and don't recall introducing myself to anyone since I was an awkward, pimply faced 15 year old at the time). Bob was exactly the same in person as he was in his letters to me – enthusiastic, generous with his knowledge and full of stories of the past. As I got to know him better I was able to hear all sorts of stories that never made it into print but which "filled in the blanks" with certain events and personalities. To this day I regret not buying a tape recorder, taking him out to dinner and saying "Bob, just start talking..." It helped that Bob and I had very similar collecting interests, of course. The first coin I ever bought was a US Large cent (at a cost of 75 cents, and I still have it). The second coin was a 1787 Connecticut copper, Miller 39.1-h.1, a nice VF piece that cost around $35, a veritable fortune at the time (I was then 7 or 8 and was flush with birthday cash and visited a coin store near where my family had dinner to celebrate – fortuitous timing indeed). From that moment on, Connecticut coppers were one of my main areas to collect, along with Washingtonia (a collection I soon traded to the late Jack Collins, igniting his passion for the series, and getting some rare books in return, including an original Dr. Hall...on 1787 Connecticut coppers, of course). Bob and I were soon corresponding every few months or so. He answered questions patiently, pointed me in the right directions for further research and would occasionally mail surprises out of the blue – some photographs, copies of the plates he had done, copies of notes he had made, whatever. Perhaps the biggest surprise was when a large Registered package arrived – Bob sent me what was left of his third collection of Connecticut coppers; perhaps 100-odd coins (the first two had gone to Ned Barnsely and Ted Craige respectively, and George Perkins had the pick of the third collection before I did). Imagine today a dealer sending a kid – and one he hadn't yet personally met! – about $5,000 in coins, which would be the equivalent of about $50,000 today. There were a bunch I needed, of course, and I finally ended up interested in about half of the box. I didn't even need to explain my finances to Bob, he said "pay me what you can, as you can." It took a year or so to get it all paid, but Bob's kindness stuck with me, and I've always offered collectors the same type of payments without interest. Perhaps it was just good business sense on Bob's part – get some new blood, and get them hooked! – but it was far more his generosity, kindness and his faith in collectors. Bob soon got me addicted to my next major area of collecting, the French Colonies coinage for North America. For well over a decade I was lucky enough to get first pick of his duplicates (a privilege that lasted until C4 Editor Syd Martin also became interested in the series, and then we were both shown things at the same time, often splitting them between us). Bob's ongoing research and addictive enthusiasm meant that I went from doing a mintmark set (one from each of the 28 French mints), to a date and mintmark set of 100-odd pieces to getting interested in die varieties and other adjunct areas and eventually forming not one, but two collections that were over 800 coins apiece. Sharing my new finds with Bob was always a thrill – he got as excited as a kid when something new was found (and I hope that we ALL retain that kind of childlike wonder well into our own sunset years). Indeed, in one of his last typed letters to me (he eventually switched to e-mail, one of the first "seniors" I knew who could figure it out) he said that the photograph he picked for his profile in his French Colonies book was him sitting at his microscope and examining a box of coins – which was actually my collection that I had mailed him intact for him to go through to find new varieties! Not unexpectedly Bob’s excellent eyes found a good dozen-plus new additions to his Sous Marques section in that box – and he good-naturedly cursed me for forcing him to renumber all the varieties he already had just to get them in. Bob was a fixture at coin shows for decades, hitting many of the larger ones in the northeast, as well as some of the FUN shows after he retired to Florida fulltime. He set up at every C4 Convention in Boston until his health declined too much to do so (though he always held out hope that he would be able to set up the following year, something that sadly never happened). Despite having a room full of colonials, Bob's table was ALWAYS my first stop, facing the entrance door, slightly to the left as you walked in. Some years there would be lots of additions to my collection, other years just a few – but there was never a show that he didn't have something that called out to me. One needed to get to his table fast, because he would invariably have a crowd 3 or 4 people deep – some looking, some asking questions, some reminiscing, all enjoying their time with a very special man. But one never mistook his kindness for weakness of any sort – I remember one ANA convention where Bob had brought a very large selection of choice colonials. Don Partrick who, even then, had the finest collection of colonials formed had put together a large pile of things he wanted to buy, in the tens of thousands of dollars. He wanted a sizable discount, 20% if memory serves. Bob looked at him and said "Don, I'm a 10% kind of guy and I'm not giving you more off than that." These two icons stared at each other a bit and Don pulled out his checkbook and bought the group. Had he held out for 11%, Bob would have let him walk away! At another ANA show a dealer had set up with a display of colonials that included a large number of counterfeits in off-metals, phony die trials and the like. One came with a purported letter from Bob, claiming that a St. Patrick die trial was absolutely genuine – the letter was a poor Xerox with every other word misspelled, and Bob who was quite the literate man was furious when I mentioned it to him. He ran over to that table and demanded the letter be given to him where he ripped it up and threw it at the dealer, saying that his integrity was never to be soiled like that again. While many collectors today know of other giants from the 50's and 60's only from their written works or the (eventual) sale of their collections – Ford, Newman, Picker, Hoch, Keller, Spilman, Breen, etc. – Bob was more than just a name that they read about. He was out there, meeting people, fueling their enthusiasm and taking care of his own intellectual curiosity along the way. Bob was a lover of the unexplored and a keen student of minutiae. When you look at some of the areas that he published monographs on – the 1783 Washington coinage, the 1767-A French Colonies Copper sols, the Atlee "Machin's Mills" Coppers and the billon coinage for the French Colonies – you see the workings of a mind who could make some sort of sense of areas that were previously ignored, or thought too complex to study seriously. Most of this research was, of course, done the old fashioned way – hands on examination of actual coins, long before the ease of digital files could be quickly zapped around the world, with descriptions meticulously written out by hand. But Bob also did significant research that was never published, but which served as the basis for other researchers. Syd Martin's works on Rosa Americana, Hibernia and his soon-to-be-published work that includes the 1721-2 French Colonies copper 9 Deniers all owe a debt of gratitude to the groundbreaking work Bob did in those areas – and unlike some collectors of his generation, notably John Ford, Bob was always willing to share his information with other collectors, not just hoard it for his own use. Bob's French Colonies collection, which was sold by Stack's shortly after his book on the series came out, may be the only public auction with his name on the coins sold. But Bob actually formed many first-rate collections that were dispersed privately, and often purchased intact by serious collectors. His Massachusetts silver was the basis of the Hains collection, his Massachusetts coppers went to a private collector, as did his Vermont coppers, his Atlee coppers, New York patterns, Washingtonia and other fine groups he formed at a time where there was very little real competition. He put together excellent collections of world coins, paper money, advertising paper (which he also wrote the standard reference book on) and, like most of us, also collected outside of numismatics, especially some areas of antiques – and, not unexpectedly, he wrote about those areas too, authoring a work on "head vases" after he formed one of the largest collections ever put together (these were vases in the form of a head, often a famous person, which were popular in the mid-20th century, though some went back to the 19th century). Bob always did well when he sold his collections – not only because he owned things for long periods of time, but because he had the knowledge and skill to cherrypick rarities, had contacts going back decades that supplied him with goodies and collectors who trusted his judgment on what things were worth, a trust that was well-earned over the course of his career. He was the consummate gentleman dealer, never holding out for absolute top dollar on every item he sold (or worrying if someone slabbed a coin 1 point better than his). Collectors today rightfully prize anything with a Vlack pedigree – especially if they come in some of his older custom-made coin envelopes, the finest of which show a witch on a broom in red ink (the image taken from a “We All Have Our Hobbies” token), with the details of each coin painstakingly typewritten, often in a distinct green ink. Colonial numismatics has been fortunate in attracting some very intelligent and enthusiastic people, but in this writer's opinion there are four that contributed far more than the usual level of participation. There were many others, of course, some who formed great collections but didn’t participate much other than the actual acquiring; some who wrote an occasional article or made a nice discovery, and others who participated for short periods of time and then left. But these "Big Four" did more than all the rest to popularize colonials – and without them, the hobby as we know it would be a vastly different area. Those four are Eric Newman, Walter Breen, Jim Spilman and Bob Vlack, and we are all better off for much of their interests being focused on areas we collect. Bob was one of the last of a dying breed, a combination of collector, researcher, writer and dealer, something of a Renaissance man since he did all these areas equally well (none of the other three ever really adding dealer to their resumes). I was most fortunate to know him for such a long period of time and to call him a friend. To read the earlier E-Sylum article, see:

Wayne Homren, Editor The Numismatic Bibliomania Society is a non-profit organization promoting numismatic literature. See our web site at coinbooks.org. To submit items for publication in The E-Sylum, write to the Editor at this address: whomren@gmail.com To subscribe go to: https://my.binhost.com/lists/listinfo/esylum All Rights Reserved. NBS Home Page Contact the NBS webmaster 
|